Best Microbial Books:
There are several books available that cover various aspects of microbiology, including microbial testing, microbial limit testing, and related topics. Here are a few recommendations:
Microbial Limit Test
The term is most commonly used in the context of bioburden testing, also known as microbial limit testing, which is performed on pharmaceutical and medical products for quality control purposes. Microbial levels in pharmaceutical or medical products must be controlled during processing and handling. Bioburden or microbial limit testing on these products demonstrates that these requirements have been met. Bioburden testing for medical devices manufactured or used in the United States is governed by Title 21 of the Code of Federal Regulations, and worldwide by ISO 11737.
The goal of bioburden testing is to determine the total number of viable microorganisms (total microbial count) on a medical device prior to final sterilisation before implantation or use.
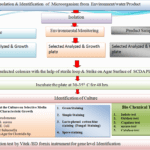
Flow Diagram of Microbial Limit Test:-
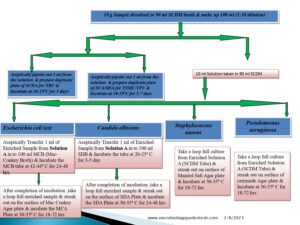
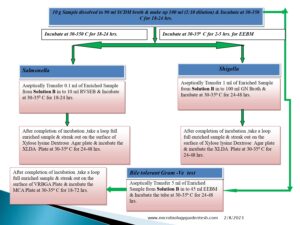
Pingback:-


I¦ve recently started a website, the information you offer on this site has helped me greatly. Thank you for all of your time & work.